Author’s note: This article is not intended to give medical advice; it is a piece of work presenting research and ideas that may be of interest to both patients and clinicians. Any change in diet must be overseen by your Oncology team, along with allied health professionals who have expertise in the area.
This article has been researched, written, and edited by the author. No part of this article has been researched, written, or edited using artificial intelligence. It is of utmost importance for the author to continue to provide authentic research articles. The author has reviewed, read, and provided academic references for this piece, which are available at the end.
Introduction
One of the most common and distressing side effects that I see in clinical practice is adverse effects on the gastrointestinal tract that are brought on by radiation and certain chemotherapeutic drugs. Herein, this condition will be referred to as ‘treatment-induced gastrointestinal injury [TIGI]. Chemotherapy and radiation are life-saving to many people; however, conventional medicine has not yet recommended integrative treatments that could both prevent and treat TIGI. The following article provides a brief overview of the types of treatments available, the ways in which treatment damages the GI tract, and discusses how the low FODMAP diet may be useful in this context.
The idea of this article was based on the review of a recent paper by Arwa Almasaudi titled “A review of the efficacy of the low fermentable oligosaccharides, monosaccharides and polyols (FODMAP) diet in managing symptoms related to cancer treatment’ (1). This is a current review summarising the evidence to date for the use of low FODMAP in TIGI.
What symptoms occur from cancer treatment, and why
Cancer patients often encounter gastrointestinal challenges due to symptoms associated with the disease itself or the side effects of treatments such as radiation and chemotherapy. It is well known that chemotherapy and radiotherapy exert toxic effects on the intestinal mucosa (2). It is estimated that 40 % of patients undergoing standard-dose chemotherapy and 60% to 100% of patients undergoing high-dose chemotherapy will suffer from gastrointestinal toxicity (2). In addition to this, 50% of patients who have undergone abdominal/pelvic radiation will also experience adverse effects (3). As a result of this damage, ongoing inflammation leads to long-term complications, including scarring, strictures, and fistulas that compromise normal intestinal function (4).
Patients undergoing treatments that lead to TIGI can show similar symptoms to a person suffering from irritable bowel syndrome [IBS]. To illustrate, a person suffering from IBS and another person suffering from treatment-related injury will both experience symptoms such as diarrhea, stomach cramps, dehydration, malnutrition, bloating, and altered bowel habits (2,5), resulting in a lowered quality of life. Could this mean that effective treatment for IBS could also be applied to TIGI injury? Regrettably, current treatment options for TIGI are limited, leaving a significant unmet need for effective management. To add another layer of complexity, cancer survivors are often encouraged to follow general lifestyle recommendations with potential beneficial effects on general health and recurrence of the disease, without recognising that such advice may negatively affect bowel symptoms (6). For example, while a high-fibre diet is beneficial for a healthy gut, it may worsen gastrointestinal symptoms when the gut is damaged. This is where the important need for personalised care should be considered.
How Does the Injury to the Gastrointestinal Tract Occur?
The mechanisms by which some cancer treatments damage the intestine are complex and remain an active area of research. The following section provides a brief overview of how chemotherapy and radiation therapy may affect intestinal function. Knowing this allows insight into how these side effects may be prevented and treated.
Chemotherapy
Although the mechanisms by which certain chemotherapy drugs injure the gastrointestinal tract are not fully understood, substantial research in this area suggests that improved treatments may become available once the exact causes are identified. Chemotherapeutic agents are designed to kill rapidly dividing cells, such as cancer cells; however, this also means they target the fast-dividing cells in the gastrointestinal tract. The chemotherapeutic drugs most likely to cause gastrointestinal toxicity include 5-FU, capecitabine, methotrexate, irinotecan, cyclophosphamide, and cisplatin (2). As a result of these drugs, gastrointestinal tract damage occurs, leading to significant changes in the gut microbiota and an increased incidence of dysbiosis (leaky gut) (2).
The healthy gut is lined with a special structure called ‘villi’. These villi house specialised cells that produce substances that form a protective coating of the gut, helping it stay healthy and intact. Unfortunately, when these villi are harmed, it also affects the person’s ability to absorb nutrients. This intestinal damage impairs the gut’s ability to digest dietary carbohydrates, leading to adverse gastrointestinal symptoms (7). This is an important consideration, as it provides a rationale for considering a low FODMAP diet.
Radiation
While chemotherapy is well known to affect the gut, radiation therapy can also cause intestinal damage and gastrointestinal dysfunction. Radiation-induced intestinal injury is one of the most common complications caused by radiotherapy for pelvic and abdominal tumors (3). The intestine can receive either direct or indirect injury. Direct injury occurs when the intestine is directly in the radiation field, whereas indirect injury results from the intestines occupying substantial volume and therefore not being fully shielded, leading to accidental exposure (3). Although both the small and large intestines are affected, the small intestine is particularly vulnerable to radiation exposure due to its rapid cell turnover (4). Radiation damages intestinal stem cells, which give rise to several specialised cell types that maintain healthy intestinal function (4). When these stem cells are damaged, they are unable to produce the essential cells that maintain gut health and protection.
It is widely recognised that radiation leads to varying degrees of intestinal dysfunction, leading to side-effects that include vomiting, weight loss, anorexia, dehydration, diarrhoea, and infections (3). Early injury from radiation can occur within four hours of the first radiation dose, with most of the acute injury typically occurring within three months after treatment. Chronic injury can develop more than three months post-radiation and also manifest years after radiation has finished (4).
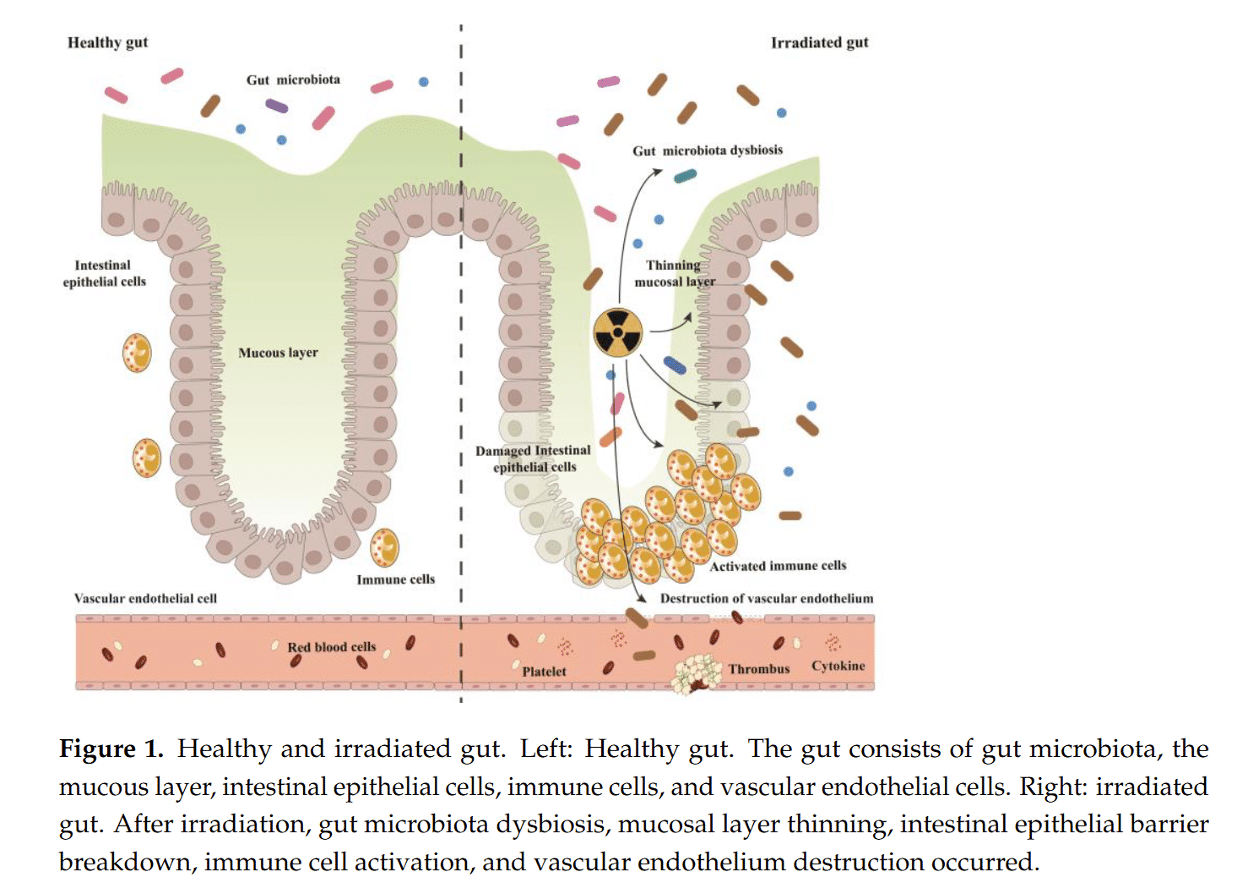
Diagram credit (3)
What is the standard treatment for TIGI?
The treatment of TIGI after cancer treatment aims to address symptoms; however, prevention and guidance on nutritional management are severely lacking (3). Currently, the approved strategies to manage GI issues from anticancer treatments are aimed at reducing diarrhea and cramping. In conventional oncology, first-line treatment typically involves loperamide, which is a medication that reduces intestinal motility, thereby reducing diarrhea and cramping. The second-line treatment, Octreotide, is generally used when loperamide fails. By mimicking a natural hormone that reduces water in the gut and also reduces the intestines’ contractions, therefore reducing diarrhea and cramping.
What is FODMAP
Low FODMAP diets may help minimise adverse GI events associated with chemotherapy and radiation (1). FODMAP stands for fermentable oligosaccharides, disaccharides, monosaccharides, and polyols, which are groups of carbohydrates that, when present in the diet, can contribute to symptoms of irritable bowel syndrome. Foods high in FODMAP carbohydrates are poorly absorbed in the small intestine; when they reach the large intestine, they are fermented by gut bacteria, producing gas and drawing water into the intestine, thereby creating a laxative effect. In susceptible people, this process can cause symptoms such as bloating, abdominal pain, diarrhea, gas, and altered bowel habits, which are the same symptoms found in patients with gastrointestinal damage from anticancer treatments. It therefore makes sense that reducing FODMAP intake would benefit patients experiencing adverse effects from their treatment.
What is low FODMAP
The low FODMAP diet involves temporarily restricting high-FODMAP foods, followed by a structured reintroduction to assess the individual’s tolerance. The goal of the FODMAP diet is to reduce symptoms while maintaining a nutritionally balanced and varied diet. Evidence suggests that consultation with an experienced dietitian ensures safe practices, adherence, and outcomes. This diet is not recommended for long-term use; therefore, working with an expert ensures that this treatment is used only when necessary (8). The gut microbiome relies on dietary diversity, including FODMAP foods, so reintroduction is also important. A dietitian should have the skills to guide the patient through specific phases while monitoring adequate nutritional intake.
The Research
Below is a summary of four papers that featured in the review by (1). These recent academic papers present various oncology scenarios in which a low-FODMAP diet has demonstrated efficacy.
1. Low FODMAP for the prevention of radiation injury in prostate cancer
One particularly noteworthy study is by (9), who examined patients undergoing radiation therapy for prostate cancer. When patients undergo radiation for prostate cancer, the intestines and especially the rectum often fall within the field of radiation, resulting in early and late side effects (10). The author hypothesised that implementing a low-FODMAP diet would minimise prostate movement during radiation by reducing rectal gas and volume, both of which are known to increase injury risk. The low FODMAP diet was followed during radiation in the experimental group, with the control group following their usual diet. The study found that rectal gas volume and rectal gas were considerably reduced during radiation, suggesting that a low FODMAP diet may be beneficial in reducing rectal toxicity during radiotherapy. This pilot study may lay the groundwork for future research that may establish low FODMAP as part of standard radiation protocols.
2. Low FODMAP during chemotherapy for colorectal cancer
(7)and colleagues examined the correlation between consuming FODMAP and the severity of GI symptoms in patients receiving adjunct treatment of 5FU for colorectal cancer. In colorectal cancer, gastrointestinal toxicity is particularly prevalent due to the target tissue being within the gastrointestinal tract (11). The participants were asked to keep a 4-day food diary and record any symptoms experienced during treatment. Upon completion of the diary, the foods were then categorised into high- and low-FODMAP groups. Not surprisingly, the study indicated that patients with high consumption of FODMAP-rich foods were more likely to experience diarrhea than those with low consumption. The study concluded that there was a significant association between total FODMAP intake and diarrhea symptoms (7)
3. Low FODMAP and radiation-induced irritable bowel syndrome
Larsen and colleagues conducted a small pilot study on females who were suffering from radiation-related irritable bowel syndrome symptoms due to treatment for gynecological cancer. These participants followed a low FODMAP diet for four weeks. As a result of the modified diet, there was a marked reduction in symptoms, as shown in the table below. Two participants in the study achieved complete remission of their gastrointestinal symptoms (12)
(13) conducted a randomised trial comparing FODMAP with a control group consuming a typical Mexican diet. The participants were undergoing external-beam radiation for cervical cancer. The conclusion of the study determined that low FODMAP was a low-cost, high-performance measure that reduces end-of-treatment quality of life and symptom deterioration (13)
Conclusion
The use of the low FODMAP diet may benefit patients with TIGI. Although much research points to a positive effect, more robust trials are needed in this area. The use of an expert health professional is critical when altering the diet in persons with cancer due to the risk of weight loss and nutritional deficiencies. Overall, this is an exciting area for consideration and development.
Reference List
- Almasaudi AS. A review of the efficacy of the low fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAP) diet in managing gastrointestinal symptoms related to cancer treatment. Cureus [Internet]. 2024 Mar;16(3):e56579. Available from: http://dx.doi.org/10.7759/cureus.56579
- Akbarali HI, Muchhala KH, Jessup DK, Cheatham S. Chemotherapy induced gastrointestinal toxicities. Adv Cancer Res [Internet]. 2022 Mar 21;155:131–66. Available from: http://dx.doi.org/10.1016/bs.acr.2022.02.007
- Lu Q, Liang Y, Tian S, Jin J, Zhao Y, Fan H. Radiation-induced intestinal injury: Injury mechanism and potential treatment strategies. Toxics [Internet]. 2023 Dec 10;11(12):1011. Available from: http://dx.doi.org/10.3390/toxics11121011
- Hu Z, Zhang J, Li H, Wang X, Zhang G, Cui H, et al. Research progress on the hallmarks of radiation-induced intestinal injury: Mechanisms, biomarkers and therapeutic targets. Arch Biochem Biophys [Internet]. 2025 Oct;772(110562):110562. Available from: http://dx.doi.org/10.1016/j.abb.2025.110562
- Black CJ, Ford AC. An evidence-based update on the diagnosis and management of irritable bowel syndrome. Expert Rev Gastroenterol Hepatol [Internet]. 2025 Jan 21;1–16. Available from: http://dx.doi.org/10.1080/17474124.2025.2455586
- Borre M, Fassov J, Poulsen JL, Christensen P, Laurberg S, Drewes AM, et al. Dietary intervention improves gastrointestinal symptoms after treatment of cancer in the pelvic organs. J Clin Med [Internet]. 2023 Jul 19 [cited 2026 Feb 2];12(14):4766. Available from: https://www.mdpi.com/2077-0383/12/14/4766
- Holma R, Laatikainen R, Orell H, Joensuu H, Peuhkuri K, Poussa T, et al. Consumption of lactose, other FODMAPs and diarrhoea during adjuvant 5-fluorouracil chemotherapy for colorectal cancer. Nutrients [Internet]. 2020 Feb 4 [cited 2026 Jan 29];12(2):407. Available from: https://www.mdpi.com/2072-6643/12/2/407
- Tuck CJ, Reed DE, Muir JG, Vanner SJ. Implementation of the low FODMAP diet in functional gastrointestinal symptoms: A real-world experience. Neurogastroenterol Motil [Internet]. 2020 Jan;32(1):e13730. Available from: http://dx.doi.org/10.1111/nmo.13730
- Schaefer C, Zamboglou C, Volegova-Neher N, Martini C, Nicolay NH, Schmidt-Hegemann NS, et al. Impact of a low FODMAP diet on the amount of rectal gas and rectal volume during radiotherapy in patients with prostate cancer – a prospective pilot study. Radiat Oncol [Internet]. 2020 Jan 30;15(1):27. Available from: http://dx.doi.org/10.1186/s13014-020-1474-y
- Rombouts AJM, Hugen N, van Beek JJP, Poortmans PMP, de Wilt JHW, Nagtegaal ID. Does pelvic radiation increase rectal cancer incidence? – A systematic review and meta-analysis. Cancer Treat Rev [Internet]. 2018 Jul;68:136–44. Available from: http://dx.doi.org/10.1016/j.ctrv.2018.05.008
- Han CJ, Ning X, Burd CE, Spakowicz DJ, Tounkara F, Kalady MF, et al. Chemotoxicity and associated risk factors in colorectal cancer: A systematic review and meta-analysis. Cancers (Basel) [Internet]. 2024 Jul 20 [cited 2026 Feb 5];16(14):2597. Available from: https://www.mdpi.com/2072-6694/16/14/2597
- Larsen T, Hausken T, Otteraaen Ystad S, Hovdenak N, Mueller B, Lied GA. Does the low FODMAP diet improve symptoms of radiation-induced enteropathy? A pilot study. Scand J Gastroenterol [Internet]. 2018 May 4;53(5):541–8. Available from: http://dx.doi.org/10.1080/00365521.2017.1397186
- Soto-Lugo JH, Souto-Del Bosque MÁ, Vázquez-Martínez CA. Effectiveness of nutritional intervention in reduction of gastrointestinal toxicity during external beam radiotherapy in women with gynecological tumors. Gac Mex Oncol [Internet]. 2022 Sep 7;16(2). Available from: http://dx.doi.org/10.24875/j.gamo.17000016




